Protaryx Medical, a Maryland-based company dedicated to “minimally invasive left-heart interventional therapies,” announced that its Transseptal Puncture Device received 510(k) clearance from the FDA.
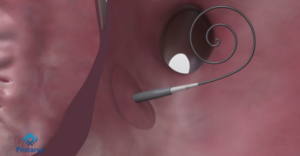
Transseptal puncture is designed to enable access to the left side of the heart, which allows physicians to perform “catheter-based” treatments such as “atrial fibrillation (AF) ablation, left atrial appendage (LAA) closure, and mitral valve repair/replacement (TVMR or TEER).”
The Protaryx Transseptal Puncture Device enables physicians to reach the left atrium without needing to repeatedly change the catheter or sheath. Protaryx further advertises its Transseptal Puncture Device as having an “extendable, highly echogenic atraumatic probe” as well as “incorporat[ing] a proprietary RF guidewire technology compatible with standard electrosurgical generators.”
Protaryx’s CEO, David Mester, referred to receiving 501(k) clearance as a “defining moment for Protaryx” that “reflects the dedication of [his] team to solving one of the most critical challenges in safe and efficient transseptal access procedures.” Dr. James Gammie, Protaryx’s cofounder and the System Chief of Cardiac Surgery at Johns Hopkins, added: “By simplifying access to the left atrium, the device can expand procedural adoption while improving safety, precision, and ease-of-use.”
This news comes as the value of global transcatheter heart valve replacement market is expected to reach $19.62 billion by 2026, with North America dominating the global market.
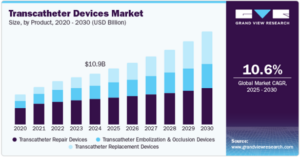
There accordingly appear to be major commercial opportunities in the transcatheter devices market. However, procedural risks, such as obtaining regulatory approval, are expected to remain a challenge.
Tags
510(k) Clearance, 510(k) Premarket Notification, afib, atrial fibrillation, atrial therapy, catheter, hear, Left Atrial Appendage (LAA), mitral valve repair, Protaryx, Transcatheter Mitral Valve Replacement (TMVR), transseptal, transseptal punch, transseptal puncture device, transseptal punture, U.S. Food & Drug Administration (FDA), zero exchange
